This will be a short post, because while I found this fact to be a revelation, I suspect most people already know this intuitively, and to them, I sound pretty goddamn stupid.
It’s against policy to sound stupid in the public sphere, but I risk broadcasting my density here for the benefit of those who, like me, have always insisted on shaking the batches before every use.
Batching:
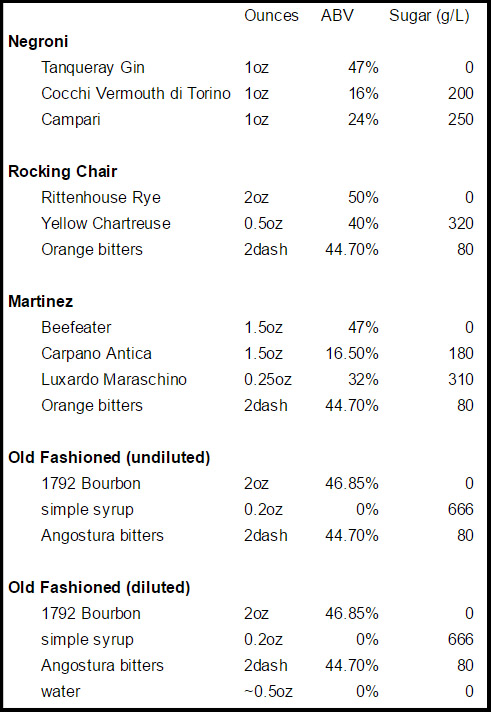
In a busy bar, you’ll often batch liqueurs, syrups, or even base spirits together to make service more efficient, turning a 5 pour drink into a 2 or 3 pour drink. Thus, for something like the Mane of Needles, say:
2oz Rye — becomes — 2oz Rye
0.75oz Carpano Antica 1.75oz batch
0.5oz Campari dash orange bitters
0.25oz Benedictine
0.25oz Fernet
dash orange bitters
Easier, no? I’ve been a fan of batching for a long time, but I’ve always warned my bartenders: agitate the batches if it’s the first time you’re touching them that day. It only made sense to me that the constituent ingredients would settle out over time, the way juice does.
My intention was to figure how quickly they separated, not whether they did at all, so I set up a couple experiments. I made 5 cocktails with ingredients of dramatically different sugar levels, ABV, and colors (all that data is at the bottom. if you’re curious) sealed them in glass bottles, and tucked them away.
![[bi] initial](https://drinksanddrinking.com/wp-content/uploads/2016/09/bi-initial.jpg?w=676&h=418)
The general idea was to check every few hours to see how quickly the colors separated. My hypothesis was that it would happen within 6-8 hours.
First few hours: nothing. First few days: nothing. After two weeks, they still looked like this:
Convinced I was missing some minute but crucial gradient, I held it up to bright light and looked harder:
Zero color variation.
“Ok,” I thought, “maybe whatever accounts for color completely mixes in but something must settle out, right?” So I took a siphon, and siphoned off each bottle into three glasses: top 1/3rd, middle 1/3rd, bottom 1/3rd. And tasted them all side by side. And nothing. They’re exactly the same.
Do the ingredients really not separate over time?
Obvious Answer:
Of course they don’t separate over time.
If alcohol and water settled out, a bottle of vodka would be stronger on top than it is on the bottom. If sugar settled out of alcohol and water, a bottle of Campari would be sweeter on the bottom than on top.
When you first add things together, they’re not completely mixed, and you see sugary wisps in the liquid. But shake or stir it a couple times, and once those wisps go away — once it’s all fully mixed together — your job is done. The liquid doesn’t know it used to be 3 different things. All it knows is that is has a certain amount of water, sugar, and alcohol, and because of Brownian Motion, the levels thereof will be constant, throughout the liquid, until the end of time.
Again, this may be obvious to you. It may seem like I’m urgently tapping you on the shoulder to tell you that giraffes are tall. And in hindsight, yeah, of course. But it took me 2 weeks of experiments and a long text conversation with a friend who has a Ph.D. in chemistry (thanks, Addison) to work it out. So, you know. There’s that.
Bonus Fact:
Solids, of course, settle in the bottom of the bottle. In bottled citrus juice, the pulp starts to settle within the hour. But what about bitters?
Angostura bitters does indeed have tiny solid particulate matter that settles out over time. Look closely to the bottom of the bottle:
Clearer still is a brief close-up video to see them dancing about:
I couldn’t possibly tell you how much of an effect those little particles have on flavor. I have no idea. But it’s probably a good idea to not batch your bitters, and to add them à la minute to each drink.
Added Bonus Fact:
This principle has an appealing corollary, which is that if you, say, found an ideal gin and sweet vermouth for a Negroni, you could just pre-batch a bottle of negroni and keep it at home for easy cocktails after long shifts. Food for thought.
Experiment Data:
This wasn’t interesting enough to put in the body, but in case your curious, these are the cocktails I chose because the ingredients had widely disparate sugar & alcohol levels, and were of different colors. Sugar levels are taken from educated guesses by smarter people than myself, most notably Dave Arnold in Liquid Intelligence and this random, helpful little website.
![[bi]two weeks later](https://drinksanddrinking.com/wp-content/uploads/2016/09/bitwo-weeks-later.jpg?w=676)
![[bi] montage](https://drinksanddrinking.com/wp-content/uploads/2016/09/bi-montage.jpg?w=676&h=344)
![[bi] bitters sediment](https://drinksanddrinking.com/wp-content/uploads/2016/09/bi-bitters-sediment.jpg?w=676)
I love your blog and approach to cocktails. When entertaining large crowds I always make batched drinks. But I never have to make them very far in advance. I do have a few questions. Do I have to refrigerate a batched cocktail the uses infused simple syrups? And do you wait to add any citrus used in the cocktail till the last minute? Thanks in advance.
Thanks Jason for writing this article (I know it was 6 years ago) but it was the exact information I was looking for.
Very happy to help, sir. I’m glad it wasn’t just me.
I know this article is old, but curious if you can help! I’m trying to make a liqueur with juice from seaberries; I would have done the infusion with the actual berries, but all I was given was the freshly-pressed juice. The juice is fully liquid, but it’s a foamy/opaque juice.
I pour 2x vodka into the juice, planning to let it sit for a month and then add simply syrup to make a liqueur… but the juice and alcohol are separating! When I stir them, there’s no pulp or pull (they stir together easily), but as soon as I stop stirring, a few minutes later the juice sits fully on top with the vodka at the bottom. Is this just a totally doomed project?
Hi Anonymous person —
I don’t know what a Seaberry is, so there may be some special thing I’m not considering, but as a rule of thumb:
The vodka shouldn’t be separating out. You can certainly layer things by density, but once mixed, it should be mixed forever — or, better, the things that mix forever (are “miscible”) are at least alcohol, water, sugar, and I believe acidity. If it’s good and mixed, it should stay that way. If you’re experiencing something layering out of the mixture, it could be pulp, or foam(?), or some other aspect of the liquid, but as far as I understand this process, the alcohol, sugar, water, and acidity should be uniform throughout.
I hear you saying there’s no pulp… but I still wonder if filtration is your answer. I’m a fan of Milk Nut Bags (search on Amazon) for this kind of thing — there’s lots of ways to get a finer filter (cheese cloth, coffee filter, centrifuge) but Milk Nut Bags are strong, and you can squeeze them a bit when you’re impatient.
That may help, and it may not help at all, but that’s my best guess. Happy liqueuring.
— Jason
I’m having the same issue with watermelon/cucumber mint in a Cornelius keg
Filtration is extraordinarily important, and never more so than when pumped out of a corny keg. Filter through a superbag.
Hi Jason,
I wondered if you can help, is it okay to batch alcoholic and non alcoholic syrups/liqueurs such as apricot brandy/cherry brandy, monin syrups or grenadine with other ingredients such as gin etc?
Thanks,
Maria
Hi Maria,
Yes, it’s ok! As long as the resulting mixture is still of high enough alcohol strength, the alcohol will cure the other non-alcoholic ingredients. Unfortunately I don’t know where that line is, or how low you can go until the Monin syrups start becoming vulnerable. I suspect 5% alcohol is too low, and I’m very confident that 20% alcohol is safe. So somewhere in there is a line, or more likely a gradient, but I can’t speak to it. BUT what that means is that if you, say, wanted to batch 3 parts rum (40%) to 2 parts non-alcoholic ingredients, the resulting mixture would still be 24% alcohol, which I suspect would be safe forever.
Great thanks so much 😀
First time on your website. I was like damn he’s so rude! But then I perused the site and l like it lol. You have my kind of pf sense of humor
Hi I’m making mixed drinks like rum punch. Ingredients are .5 Oz of finest call strawberry puree mix .5 Oz finest call passion fruit puree mix 4 Oz of dailys sweet and sour mix .5 Oz of paramount amaretto and 1.5 Oz of bacardi silver.
All ingredients put in a metal shaker with ice and shook for 30 to 40 seconds. Strained pour into a glass with no ice.
Problem I’m having is after it sits in the glass for a few minutes it starts separating with clear liquid at the bottom.
Can you tell what’s happening and how to stop it.
Hi! Yes, I can help, and I’m seeing perhaps I should’ve been more specific in my post:
Sugar, water, and alcohol, when mixed, are mixed forever. This does not account for fruit purees, or any other type of plant matter that you put in your drink. Over time, the actual plant matter will separate from the liquid, much as it would if there were mint or basil in there. If you want it to not separate, you’ll need to use a strawberry & passionfruit infusion or liqueur.
Thank you for sharing, I don’t know who those “most people” are that may have thought this topic was “intuitive”, I assure you, if you are “dense”….. you’re far from alone 😂.
I mean, 9 years later and a career bartender 🙋🏻♀️ is still finding your article relevant, bravo 👏 😉.! -Jenna